PAUL® Glaucoma Implant is a glaucoma drainage device designed to treat glaucoma by regulating intraocular pressure in the patient’s eye and preventing further progression of the disease. The device is constructed entirely from medical implantable grade silicone.
- PAUL® Glaucoma Implant is a micro-sized tubed aqueous shunt for the PAUL® Stability System which is designed to regulate glaucoma or raised intraocular pressure in the patient’s eye.
- Drains fluid from the interior of the eye to the exterior of the eye.
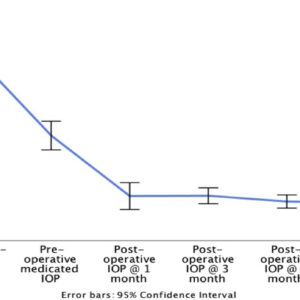
- Provides a stable& well-formed anterior chamber post-implantation in the immediate post-operation month.
- Offers effective & safe glaucoma control for the eye.
KEY NOVEL FEATURES
Micro-Sized tube
- Small Internal Calibre: Creates high flow resistance and safeguards against early hypotony.
- Small External Calibre: Occupies less space in anterior chamber and minimizes risks of tube erosion and corneal touch.
Optimized Endplate Design
- Optimal Large Plate Surface Area: More area available for aqueous filtration.
- Ideal Drainage Shape: Less filtration area covered by recti muscle.
Advanced Device Composition
- Proprietary Medical-grade Silicone: Creates a device with a new level of flexibility to facilitate the implantation process.
- Flexible Device: Less rigidity and decreases both micro-abrasion and excessive wound scarring.